ePrEP: Testing an Electronic PrEP Initiation and Maintenance Home Care System to Promote PrEP Among Adolescent MSM in Rural and Small Town Areas

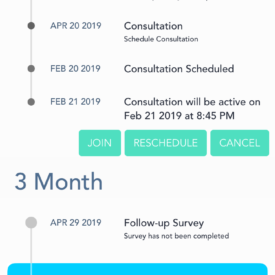
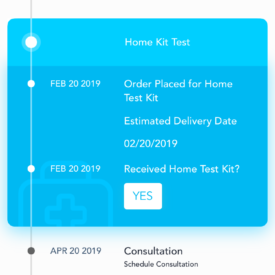
ePrEP is a home-care system for PrEP that will allow rural MSM to initiate and maintain PrEP clinical care without requiring travel to a clinician office or to a laboratory for testing. ePrEP combines a home care system for behavioral surveillance and home specimen collection/shipping for laboratory tests with app-based telemedicine and patient management.
Duration
Participants will remain in the study for 12 months after randomization
Sample Size
240 (120 intervention, 120 control).
Eligibility
- Assigned male at birth
- Age 18-29 (inclusive)
- Live in a rural or small town ZIP code based on CDC urbanicity classifications
- Laboratory confirmed HIV negative
- Owns an iOS or Android smartphone capable of running the study app
- Behaviorally indicated for PrEP
- Willing to take daily oral emtricitabine/tenofovir disoproxil fumarate (FTC/TDF) as PrEP
- Willing to use study-provided PrEP navigation services
- Willing to self-collect specimens
- More information on eligibility and exclusion criteria available on ClinicalTrials.gov
Outcomes
The primary outcome is protective levels of PrEP, as indicated by TFV-DP levels. The cutpoint used for the primary outcome measure will be TFV-DP levels considered to be a surrogate for substantial protection: >700 fmol/punch, a level indicating > 4 doses/week. Secondary outcomes will include retention in PrEP care and a number of ATN harmonized measures. We will employ standard methods of cost analyses to estimate the cost, cost-effectiveness, and cost-utility of the intervention relative to standard of care.