YouTHrive (YT) is a two-arm randomized controlled trial (RCT) to test the efficacy of an adapted version of the Thrive With Me intervention for youth living with HIV (YLWH).

YouTHrive (YT) is a 2-arm prospective randomized controlled trial (RCT) in Atlanta, Charlotte, Chapel Hill, Chicago, Houston, New York City, Philadelphia, and Tampa. To inform intervention development, we conducted 6 focus groups in Chicago, Houston, and New York and usability testing at all recruitment sites.
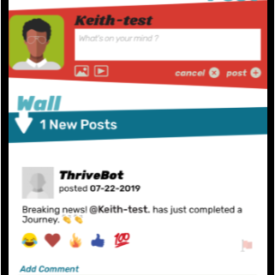
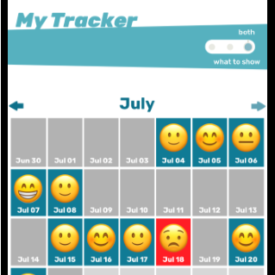
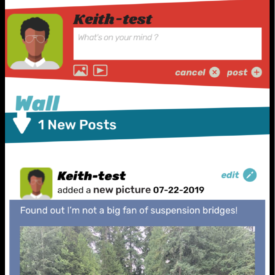
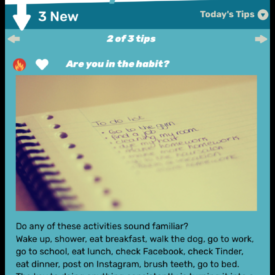
Youth randomized to the intervention condition will have access to a mobile-optimized website for 5 months that has the following features: 1) a community wall where youth can interact with each other by posting comments and replying to other participant’s comments; 2) daily rotating informational, motivational, and behavioral skills tips about living with HIV; 3) self-monitoring of HIV medications and mood; 4) and goal setting and goal tracking a suggested or a custom goal. As youth interact with the site, they will collect points and move up levels. With each new level, new features of the site are unlocked such as new avatars and color theme options.
Participants assigned to the control condition will receive a weekly email newsletter for 5 months with content about living with HIV and general well-being.
Screenshots
Duration
Participants in both the intervention and control arms are enrolled for 11 months.
Sample Size
Total study sample is 368 of which the following will be enrolled for each phase: 1) Up to 48 participants for focus groups to inform intervention adaption; 2) 12-20 participants for usability testing to finalize intervention components; 3) 300 participants for a randomized controlled trial of YT, with participants randomized to either YT (n=150) or control (n=150).
Eligibility
- 15-24 years of age at the enrollment visit
- HIV-positive status
- Residing in Atlanta, Chapel Hill, Chicago, Houston, NYC, Philadelphia, or Tampa area and available to meet with SRV staff for visits at baseline, and 5-month and 11-month follow-up assessments
- Evidence of an ART prescription with refills
- Anticipated continuous internet access and SMS messaging for the intervention period (approximately 5 months)
- Meets medical-chart verified or self-reported criteria
- More information on eligibility and exclusion criteria available on ClinicalTrials.gov
Outcomes
Primary Objective: In a 2-arm RCT (n=300), assess the efficacy of YouTHrive(YT) to sustain suppressed viral load (VL) among YLWH, compared to an HIV information-only control condition.
Secondary Objective: Assess whether YTis more beneficial for substance-using than non substance-using YLWH.